
Clinical Trial Financial Management: Investigator Grants Clinical Trial Forecasting | Medidata Solutions - Medidata Solutions

The Benefits Of Being A Principal Investigator In Clinical Research Webinar — The Clinical Trials Guru

Principal Investigator Training - Role of Principal Investigator in Clinical Research — Clinical Research Certification

Lost in Translation – What does it mean to be a “Qualified Investigator”? – Research ethics simplified™

Amazon | Investigator Initiated Trials Simplified: A Practical Guide for Clinical Trial Investigators to Conduct Iits | George, T. C. | Education & Training


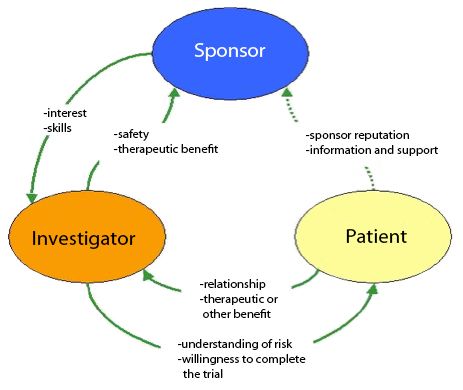




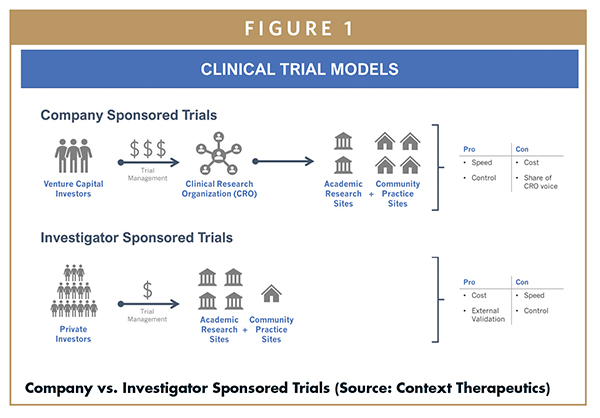







:quality(90)/)


