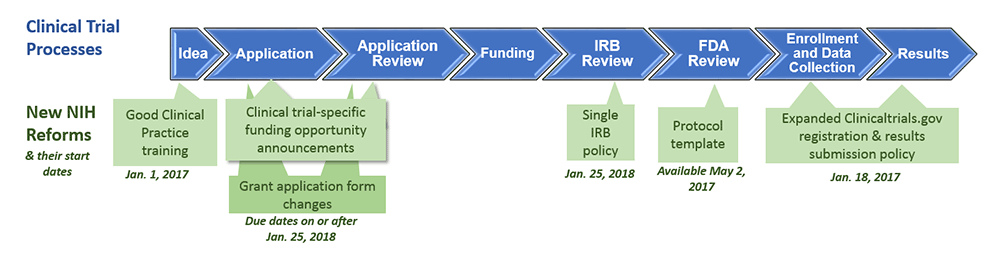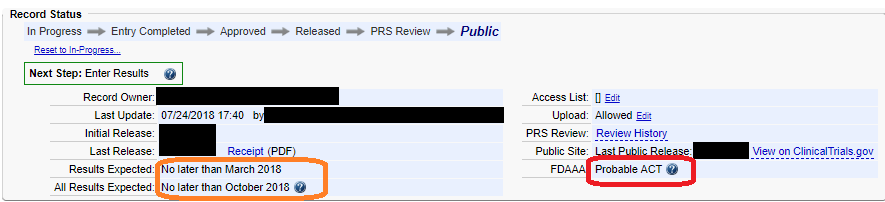
ClinicalTrials.gov Tip of the Week: Seife et al. v. HHS et al.: Results Must be Submitted for “pACTs” to Clinical Trials.gov

ClinicalTrials.gov: How to Register Your Trial - Clinical and Translational Science Institute - University at Buffalo

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet